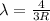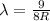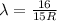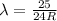Chemistry, 14.04.2020 23:42 leannaadrian
The Balmer series consists of the spectral lines from hydrogen for an electron making a transition from an excited state to the m = 2 state. The Lyman series consists of the spectral lines from hydrogen for an electron making a transition from an excited state to the m = 1 state. Determine the wavelengths of the first four spectral lines of the Lyman series (n = 2, 3, 4, and 5)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Schrodinger and heisenberg developed an alternate theory about atomic nature that contradicted some of bohr's model of the atom. how do changes resulting from new technology and evidence affect the reputation of the atomic theory?
Answers: 1

Chemistry, 22.06.2019 06:00
The tilt of the earth's axis of rotation is responsible for the a) ocean's tides. b) size of the moon. c) brightness of stars. d) earth’s seasons.
Answers: 1


Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
You know the right answer?
The Balmer series consists of the spectral lines from hydrogen for an electron making a transition f...
Questions

Mathematics, 11.07.2021 05:50


Mathematics, 11.07.2021 05:50




Business, 11.07.2021 05:50

Social Studies, 11.07.2021 05:50

Mathematics, 11.07.2021 05:50







English, 11.07.2021 06:00

Medicine, 11.07.2021 06:00


English, 11.07.2021 06:00

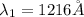
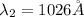
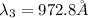
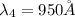
![\frac{1}{\lambda} =RZ^2[\frac{1}{n_1^2} -\frac{1}{n_2^2} ]](/tpl/images/0600/0318/c471c.png)
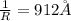 =rydberg constant
=rydberg constant