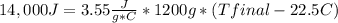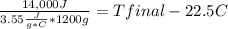Chemistry, 14.04.2020 22:50 emmaraeschool
Complete combustion of a 0.350 g sample of a compound in a bomb calorimeter releases 14.0 kJ of heat. The bomb calorimeter has a mass of 1.20 kg and a specific heat of 3.55 J/(gi°C). If the initial temperature of the calorimeter is 22.5°C, what is its final temperature? Use q equals m C subscript p Delta T..

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2

Chemistry, 22.06.2019 10:00
Drug abuse will not lead to physical and psychological dependence. true or false ?
Answers: 2

Chemistry, 22.06.2019 13:30
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1

Chemistry, 22.06.2019 21:00
Need what is special about water as a compound? how does water regulate climate? what drives water evaporation? why is the water vapor fresh water when it rises from the ocean? why might freshwater in the form of snow take longer to enter the water cycle again than liquid precipitation? what is an aquifer? what role do people play in the water cycle? plz just answer as many as you can ! thx if you !
Answers: 1
You know the right answer?
Complete combustion of a 0.350 g sample of a compound in a bomb calorimeter releases 14.0 kJ of heat...
Questions



Computers and Technology, 09.10.2020 22:01




Mathematics, 09.10.2020 22:01


Biology, 09.10.2020 22:01

History, 09.10.2020 22:01

Mathematics, 09.10.2020 22:01



Chemistry, 09.10.2020 22:01

English, 09.10.2020 22:01





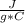 m=1.20 kg= 1200 g (1 kg=1000 g)Tfinal= ?Tinitial= 22.5 °C
m=1.20 kg= 1200 g (1 kg=1000 g)Tfinal= ?Tinitial= 22.5 °C