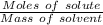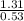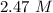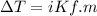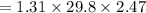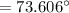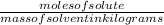Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 05:20
Asolution contains 180 g of glucose (c6h12o6) and 162 g of water. what is the mole fraction of glucose?
Answers: 3

Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 1
You know the right answer?
What is the freezing point of a solution made with 1.31 mol of CHCl3 in 530.0 g of CCl4 (Kf =29.8 de...
Questions

Mathematics, 01.05.2021 01:50

Mathematics, 01.05.2021 01:50

Mathematics, 01.05.2021 01:50

English, 01.05.2021 01:50

History, 01.05.2021 01:50

Biology, 01.05.2021 01:50

Mathematics, 01.05.2021 01:50


Mathematics, 01.05.2021 01:50



History, 01.05.2021 01:50


Mathematics, 01.05.2021 01:50



Mathematics, 01.05.2021 01:50


Geography, 01.05.2021 01:50

Chemistry, 01.05.2021 01:50

 = 29.8° C/m
= 29.8° C/m