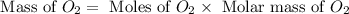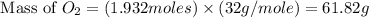Chemistry, 14.04.2020 21:10 SassyBanana
Glucose, C 6 H 12 O 6 , is used as an energy source by the human body. The overall reaction in the body is described by the equation C 6 H 12 O 6 ( aq ) + 6 O 2 ( g ) ⟶ 6 CO 2 ( g ) + 6 H 2 O ( l ) Calculate the number of grams of oxygen required to convert 58.0 g of glucose to CO 2 and H 2 O .

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
The tilt of the earth's axis of rotation is responsible for the a) ocean's tides. b) size of the moon. c) brightness of stars. d) earth’s seasons.
Answers: 1

Chemistry, 22.06.2019 12:00
Ineed this asap part i: scientific method what is the difference between science and pseudoscience? what is the scientific method?
Answers: 2


Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1
You know the right answer?
Glucose, C 6 H 12 O 6 , is used as an energy source by the human body. The overall reaction in the b...
Questions



English, 01.06.2020 14:57

Mathematics, 01.06.2020 14:57

Mathematics, 01.06.2020 14:57



Mathematics, 01.06.2020 14:57


Biology, 01.06.2020 14:57


Mathematics, 01.06.2020 14:57


English, 01.06.2020 14:57

Mathematics, 01.06.2020 14:57


Mathematics, 01.06.2020 14:57



Computers and Technology, 01.06.2020 14:57

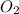 required is, 61.82 grams.
required is, 61.82 grams.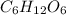
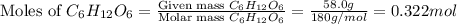
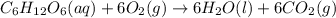
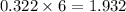 moles of
moles of