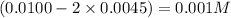Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:10
Starch and are common polysaccharide carbohydrates found in plants. sucrose glycogen fructose cellulose
Answers: 3

Chemistry, 21.06.2019 22:30
Imagine that you’re getting ready to move to a new city. when people move, they are influenced by push factors and pull factors, and you have many reasons for your move. which of the following factors is an example of a pull factor? a. wanting to move because you’ve found a great new school somewhere new b. needing to move because there are not enough resources in your old hometown c. being forced to move because your old home is gone d. having to move because there are no jobs in your current hometown
Answers: 1

Chemistry, 22.06.2019 02:30
Which element forms an ionic bond with flourine? 1) fluorine 2) carbon 3) potassium 4) oxygen
Answers: 1

Chemistry, 22.06.2019 22:00
All of the following are homogeneous mixtures except a) sugar dissolved in water. b) orange juice. c) coffee with cream. d) household vinegar. e) apple juice
Answers: 1
You know the right answer?
The reaction below is carried out at a different temperature at which Kc=0.055. This time, however,...
Questions





Computers and Technology, 17.01.2020 23:31





Mathematics, 17.01.2020 23:31



Mathematics, 17.01.2020 23:31

Mathematics, 17.01.2020 23:31



Geography, 17.01.2020 23:31



Biology, 17.01.2020 23:31

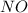 at equilibrium = 0.001 M
at equilibrium = 0.001 M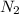 = 0.0045 M
= 0.0045 M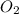 = 0.0045 M
= 0.0045 M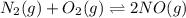
![K_c=\frac{[NO]^2}{[N_2][O_2]}](/tpl/images/0599/1341/71f8f.png)
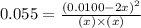
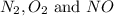 at equilibrium are :
at equilibrium are :