What is the molarity of a solution of HCl if 5.00 mL of the HCl solution is
titrated with 28.6...

Chemistry, 14.04.2020 20:43 pippyysanchezz
What is the molarity of a solution of HCl if 5.00 mL of the HCl solution is
titrated with 28.6 mL of a 0.145 M NaOH solution?
Round your answer to 3
decimal places.
HCl + NaOH → NaCl + H 2 O

Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

You know the right answer?
Questions



Biology, 13.03.2020 19:50

Computers and Technology, 13.03.2020 19:50





Mathematics, 13.03.2020 19:50

Mathematics, 13.03.2020 19:50








Mathematics, 13.03.2020 19:50


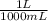 ) x (
) x (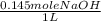 ) x (
) x (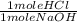 ) = 0.004147 moles HCl
) = 0.004147 moles HCl

