
Chemistry, 14.04.2020 16:12 buiratsamah
Butane (C4 H10(g), Hf = –125.6 kJ/mol) reacts with oxygen to produce carbon dioxide (CO2 , Hf = –393.5 kJ/mol ) and water (H2 O, Hf = –241.82 kJ/mol) according to the equation below. What is the enthalpy of combustion (per mole) of C4H10 (g)? Use .

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
This active feature of earth's crust in building mountain ranges as well as islands. this feature is a a) cavern. b) earthquake. c) mountain. d) volcano.
Answers: 2

Chemistry, 22.06.2019 22:30
Which process describes vaporization that takes place below the surface of a liquid? condensation melting boiling evaporation
Answers: 1

Chemistry, 22.06.2019 23:30
The density of benzene at 15 °c is 0.8787 g/ml. calculate the mass of 0.1500 l of benzene at this temperature. enter your answer in terms of grams
Answers: 2

Chemistry, 23.06.2019 06:00
If you try to move a piano and are unable to move it, did you perform any work in the scientific sense of the word? yes? or no? this question is worth 20 points! let it be correct!
Answers: 1
You know the right answer?
Butane (C4 H10(g), Hf = –125.6 kJ/mol) reacts with oxygen to produce carbon dioxide (CO2 , Hf = –393...
Questions

Mathematics, 22.09.2020 01:01

Mathematics, 22.09.2020 01:01

English, 22.09.2020 01:01

History, 22.09.2020 01:01

Biology, 22.09.2020 01:01




English, 22.09.2020 01:01




English, 22.09.2020 01:01




Mathematics, 22.09.2020 01:01



 is -2657.5 kJ
is -2657.5 kJ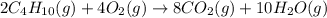
![\Delta H^o_{rxn}=[(8\times \Delta H^o_f_{CO_2(g)})+(10\times \Delta H^o_f_{H_2O(g)})]-[(1\times \Delta H^o_f_{C_4H_{10}(g)})+(4\times \Delta H^o_f_{O_2(g)})]](/tpl/images/0598/2199/66d33.png)
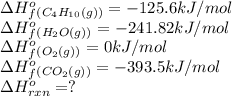
![\Delta H^o_{rxn}=[(8\times -393.5)+(10\times -241.82)]-[(2\times -125.6)+(4\times 0)]\\\\\Delta H^o_{rxn}=-5315kJ](/tpl/images/0598/2199/af1b6.png)




