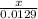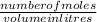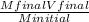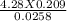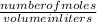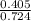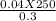Chemistry, 14.04.2020 07:44 aide1234564
1.Complete the balanced neutralization equation for the reaction below:
H2SO4 (aq) + Sr(OH)2 (aq) -->
2. How many L of a 0.209 M KI solution is needed to completely react with 2.43 g of Cu(NO3)2 according to the balanced chemical reaction:
2Cu(NO₃)₂(aq) + 4KI(aq) → 2CuI(aq) + I₂(s) + 4KNO₃(aq)
3.What volume in L of a 0.724 M Nal solution contains0.405 mol of Nal?
4. How many mL of 0.300 M NaF would be required to make a 0.0400 M solution of NaF when diluted to 250.0 mL with water?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 19:00
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1

Chemistry, 23.06.2019 10:20
Determine the mass of the object below with accuracy and to the correct degree of precision. a. 324.2 g b. 324 g c. 324.30 g d. 324.25 g
Answers: 3

Chemistry, 23.06.2019 15:40
Glucose (c6h12o6) is the simple sugar that plants make. what is the total number of atoms in glucose? 1 3 24 144
Answers: 1

Chemistry, 23.06.2019 16:00
What is a three-dimensional structure that represents the alternating pattern of particles in a crystal? a polyatomic ion a crystal lattice an ionic bond a formula unit
Answers: 3
You know the right answer?
1.Complete the balanced neutralization equation for the reaction below:
H2SO4 (aq) + Sr(OH)2 (...
H2SO4 (aq) + Sr(OH)2 (...
Questions


Mathematics, 04.05.2021 03:10

Mathematics, 04.05.2021 03:10

Geography, 04.05.2021 03:10


History, 04.05.2021 03:10

Mathematics, 04.05.2021 03:10

Mathematics, 04.05.2021 03:10

Mathematics, 04.05.2021 03:10


Social Studies, 04.05.2021 03:10

Mathematics, 04.05.2021 03:10





Mathematics, 04.05.2021 03:10

English, 04.05.2021 03:10

Mathematics, 04.05.2021 03:10

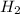 S
S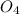 + Sr(OH)
+ Sr(OH) ⇒ SrSO4 + 2 H2O is the balanced reaction.
⇒ SrSO4 + 2 H2O is the balanced reaction.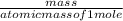
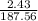
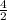 =
=