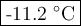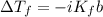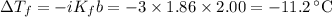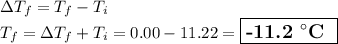Chemistry, 14.04.2020 01:51 TMeansStupidity
The normal freezing point of water is 0oC and it's freezing point depression constant is 1.86oC/m. If a 2.00 molal solution of Na2SO4 is prepared, what is the freezing point of the mixture?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3


Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1

Chemistry, 23.06.2019 08:50
Reacting masses1 calcium carbonate breaks down on heating to produce calcium oxide and carbondioxide gas.caco3 + cao + co2a student heats 15 g of calcium carbonate strongly in a crucible.relative atomic masses (a): ca = 40, c = 12, o = 16.calculate the mass of calcium oxide produced by this reaction.(5 marks)
Answers: 3
You know the right answer?
The normal freezing point of water is 0oC and it's freezing point depression constant is 1.86oC/m. I...
Questions

Mathematics, 23.07.2019 21:30



Mathematics, 23.07.2019 21:30


English, 23.07.2019 21:30




History, 23.07.2019 21:30







Mathematics, 23.07.2019 21:30

Mathematics, 23.07.2019 21:30

Mathematics, 23.07.2019 21:30