Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2


Chemistry, 23.06.2019 02:00
Which of these is a density dependent factor? a. epidemic b. earthquake c. drought d. hurricane
Answers: 2

Chemistry, 23.06.2019 03:30
In chemistry, the type of an atom (what element it is) is determined by: a) the number of protons it contains in its nucleus.b) the number of neutrons it contains in its nucleus.c) the number of protons it has in a cloud around the nucleus.d) the number of neutrons it has in a cloud around the nucleus.e) the number of electrons it exchanges with its neighbors.
Answers: 1
You know the right answer?
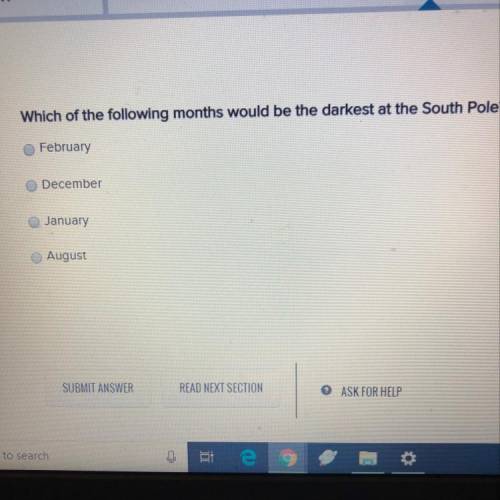
The question is in the pic plz
...
...
Questions

Spanish, 17.09.2019 19:50



History, 17.09.2019 19:50


Mathematics, 17.09.2019 19:50



Physics, 17.09.2019 19:50


Advanced Placement (AP), 17.09.2019 19:50

Arts, 17.09.2019 19:50

Chemistry, 17.09.2019 19:50



Biology, 17.09.2019 19:50

Social Studies, 17.09.2019 19:50


Mathematics, 17.09.2019 19:50

Physics, 17.09.2019 19:50