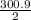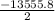Chemistry, 11.04.2020 01:50 smileyjesse6073
Calculate the enthalpy of combustion of 1 mol decane, C10H22, (l), to form CO2 and H2O. ∆Hf0 for decane is —300.9 kJ/mol using the balanced chemical equation and Appendix Table B–14.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which describes interactions between substances and stomata during photosynthesis? check all that apply. oxygen enters stomata. oxygen is released through stomata. carbon dioxide enters stomata. carbon dioxide is released through stomata. hydrogen enters stomata. hydrogen is released through stomata.
Answers: 1


Chemistry, 22.06.2019 04:00
What layer of the atmosphere is directly above the troposphere?
Answers: 1

Chemistry, 22.06.2019 16:00
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1
You know the right answer?
Calculate the enthalpy of combustion of 1 mol decane, C10H22, (l), to form CO2 and H2O. ∆Hf0 for dec...
Questions


Mathematics, 03.07.2019 21:20






History, 03.07.2019 21:20





Business, 03.07.2019 21:30


Social Studies, 03.07.2019 21:30

Social Studies, 03.07.2019 21:30




Mathematics, 03.07.2019 21:30

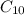
 + 31
+ 31 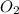 ⇒ 20 C
⇒ 20 C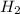 O
O