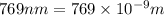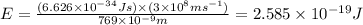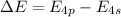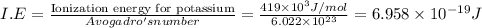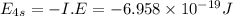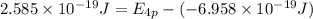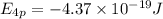Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:00
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3

Chemistry, 22.06.2019 21:20
The organs inside the body and how they function together
Answers: 3

Chemistry, 23.06.2019 00:00
In an exothermic reaction, energy may be released to the surroundings in the form of question 4 options: heat light thermal all of the above
Answers: 3

Chemistry, 23.06.2019 01:00
Which of the following is a physical change? a.burning a piece of wood b.sawing a piece of wood in half c.rust forming on an iron fence d.a copper roof changing color from orange to green
Answers: 1
You know the right answer?
The ionization energy for potassium 419 kj/mol. the wavelength of light emitted when an excited k at...
Questions


Mathematics, 27.03.2021 06:50

Mathematics, 27.03.2021 06:50




History, 27.03.2021 06:50

English, 27.03.2021 06:50


Mathematics, 27.03.2021 06:50




Spanish, 27.03.2021 06:50






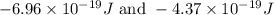
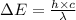
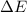 = change in energy
= change in energy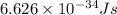
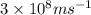
 =
=