

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 14:30
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
You know the right answer?
How many moles of O2 are produced when 1.26 moles of H2o is reacted...
Questions

English, 30.03.2021 15:20


Business, 30.03.2021 15:20

Biology, 30.03.2021 15:20


Social Studies, 30.03.2021 15:20

Social Studies, 30.03.2021 15:20



World Languages, 30.03.2021 15:20


English, 30.03.2021 15:20

Mathematics, 30.03.2021 15:20


Arts, 30.03.2021 15:20

Physics, 30.03.2021 15:20

Physics, 30.03.2021 15:20

Geography, 30.03.2021 15:30


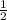 mole of O₂.
mole of O₂.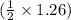 mole of O₂.
mole of O₂.

