Chemistry, 08.04.2020 21:16 elijahjacksonrp6z2o7
47.7 g chunk of ice at -58 Celsius is heated until it completely melt find the total amount of heat in joules for this process to occur. What is the total amount of energy needed overall in kilojoules (three sig figs)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:20
What would you do if you told the guy you liked that you liked him
Answers: 1

Chemistry, 22.06.2019 07:10
Remember to use the proper number of significant figures and leading zeros in all calculations.gelatin has a density of 1.27 g/cm³. if you have a blob of gelatin dessert that fills a 2.0 liter bottle, what is its mass? 2540 g2500 g3.9 x 10-43.937x 10-4
Answers: 3

Chemistry, 22.06.2019 12:30
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1

Chemistry, 22.06.2019 16:00
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1
You know the right answer?
47.7 g chunk of ice at -58 Celsius is heated until it completely melt find the total amount of heat...
Questions

Mathematics, 01.12.2020 23:30





Spanish, 01.12.2020 23:30

Chemistry, 01.12.2020 23:30





Advanced Placement (AP), 01.12.2020 23:30

Mathematics, 01.12.2020 23:30

Mathematics, 01.12.2020 23:30

Biology, 01.12.2020 23:30

Social Studies, 01.12.2020 23:30

Mathematics, 01.12.2020 23:30


Mathematics, 01.12.2020 23:30

Social Studies, 01.12.2020 23:30

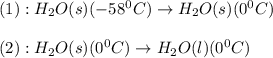
![\Delta H=[m\times c_{s}\times (T_{final}-T_{initial})]+n\times \Delta H_{fusion}](/tpl/images/0590/2015/79a71.png)
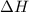 = enthalpy change = ?
= enthalpy change = ?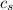 = specific heat of solid water =
= specific heat of solid water = 
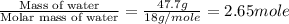
 = enthalpy change for fusion = 6.01 kJ/mole
= enthalpy change for fusion = 6.01 kJ/mole ![\Delta H=[0.0477kg\times 2.108kJ/kg^0C\times (0-(-58))^0C]+2.65mole\times 6.01kJ/mole](/tpl/images/0590/2015/2aedb.png)