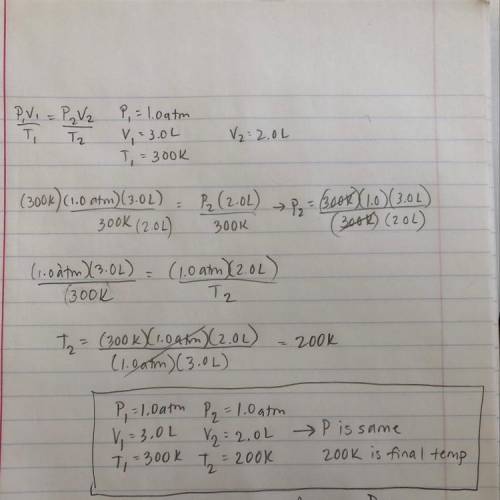Chemistry, 08.04.2020 21:09 blueval3tine
The volume of a gas is 3.0 L, the pressure is 1.0 atm, and the temperature is 300 K. A chemist changes one factor while keeping another constant so that the new volume is 2.0 L. Which of the following could be the new conditions? (5 points)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Agas occupies 475 cm^3 at 313k. find its volume at 367k. you must show all of your work to receive credit. be sure to identify which of the gas laws you will be using
Answers: 2

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1

Chemistry, 22.06.2019 21:00
One similarity and one difference between an element and a mixture of elements
Answers: 1
You know the right answer?
The volume of a gas is 3.0 L, the pressure is 1.0 atm, and the temperature is 300 K. A chemist chang...
Questions

Social Studies, 26.01.2022 04:40



Mathematics, 26.01.2022 04:40


SAT, 26.01.2022 04:40

SAT, 26.01.2022 04:40


Computers and Technology, 26.01.2022 04:40


Biology, 26.01.2022 04:40





History, 26.01.2022 04:50


Mathematics, 26.01.2022 04:50


Mathematics, 26.01.2022 04:50