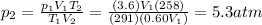Chemistry, 08.04.2020 19:24 pedroramirezr2
For many purposes we can treat nitrogen N2 as an ideal gas at temperatures above its boiling point of −196.°C. Suppose the temperature of a sample of nitrogen gas is lowered from 18.0°C to −15.0°C, and at the same time the pressure is changed. If the initial pressure was 3.6atm and the volume decreased by 40.0%, what is the final pressure? Round your answer to the correct number of significant digits.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 02:20
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 09:00
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1

Chemistry, 22.06.2019 12:20
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1
You know the right answer?
For many purposes we can treat nitrogen N2 as an ideal gas at temperatures above its boiling point o...
Questions


Social Studies, 05.10.2021 08:00

Mathematics, 05.10.2021 08:00


History, 05.10.2021 08:00



English, 05.10.2021 08:00




History, 05.10.2021 08:10

Mathematics, 05.10.2021 08:10


Business, 05.10.2021 08:10

Mathematics, 05.10.2021 08:10

Physics, 05.10.2021 08:10



Arts, 05.10.2021 08:10

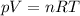

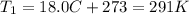 is the initial temperature of the gas
is the initial temperature of the gas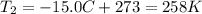 is the final temperature
is the final temperature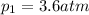 is the initial pressure
is the initial pressure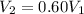 , as the volume is decreased by 40.0%
, as the volume is decreased by 40.0%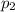 , we find the final pressure of the gas:
, we find the final pressure of the gas: