
Chemistry, 08.04.2020 18:22 fatlip2429
Calculate the change in enthalpy (LaTeX: \DeltaΔH) of a reaction with the following information:
Energy of Reactants = 129 kJ
Energy of the Products = 110 kJ
Energy of the Transition State = 198 kJ

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Asolution is made by dissolving 25.5 grams of glucose (c6h12o6) in 398 grams of water. what is the freezing point depression of the solvent if the freezing point constant is -1.86 °c/m? show all of the work needed to solve this problem.
Answers: 1


Chemistry, 22.06.2019 14:00
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
You know the right answer?
Calculate the change in enthalpy (LaTeX: \DeltaΔH) of a reaction with the following information:
Questions


Mathematics, 13.11.2020 01:00

Mathematics, 13.11.2020 01:00


Mathematics, 13.11.2020 01:00

Mathematics, 13.11.2020 01:00


History, 13.11.2020 01:00

Computers and Technology, 13.11.2020 01:00

Mathematics, 13.11.2020 01:00

Mathematics, 13.11.2020 01:00

English, 13.11.2020 01:00



Social Studies, 13.11.2020 01:00

Health, 13.11.2020 01:00

Mathematics, 13.11.2020 01:00

Biology, 13.11.2020 01:00

Arts, 13.11.2020 01:00

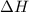 is mathematically expressed as the difference between the the total potential energy of products
is mathematically expressed as the difference between the the total potential energy of products 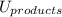 and the potential energy of the reactants
and the potential energy of the reactants 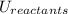 :
: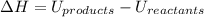
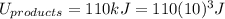
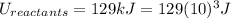
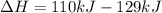
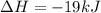 The negative sign in this result means we have a negative enthalpy, hence an exothermic reaction (where heat is released).
The negative sign in this result means we have a negative enthalpy, hence an exothermic reaction (where heat is released). 

