
Chemistry, 08.04.2020 03:35 rainbowmc6
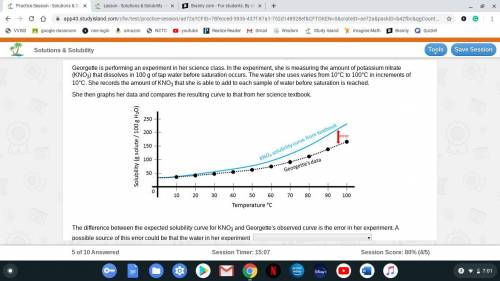
Georgette is performing an experiment in her science class. In the experiment, she is measuring the amount of potassium nitrate (KNO3) that dissolves in 100 g of tap water before saturation occurs. The water she uses varies from 10°C to 100°C in increments of 10°C. She records the amount of KNO3 that she is able to add to each sample of water before saturation is reached.
She then graphs her data and compares the resulting curve to that from her science textbook.
The difference between the expected solubility curve for KNO3 and Georgette's observed curve is the error in her experiment. A possible source of this error could be that the water in her experiment
A. did not have any substances already in it
B. already had substances dissolved in it
C. vaporized at about 40°C
D Solidified at about 40°C

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 11:30
If 4.8 moles of x and 3.4 moles of y react according to the reaction below, how many moles of the excess reactant will be left over at the end of the reaction? 3x + 2y “yields”/ x3y2. a. 1.7 mol y left over b. 1.6 mol x left over c. 0.2 mol y left over d. 0.1 mol x left over
Answers: 1

Chemistry, 23.06.2019 18:20
In a chemical reaction, the number of moles of the reactants a. should never be equal to the number of moles of the products. b. may or may not be equal to the number of moles of the products. c. depends on the amount of product that is formed. d. should always be equal to the number of moles of the products.
Answers: 3

Chemistry, 23.06.2019 23:10
An object was measured by a worker as 14.6cm long, however, the manufacturer specifications list the length of the object at 14.4cm. what is the percent error in the worker's measurement?
Answers: 1
You know the right answer?
Georgette is performing an experiment in her science class. In the experiment, she is measuring the...
Questions


Mathematics, 28.11.2020 01:00


Social Studies, 28.11.2020 01:00


Mathematics, 28.11.2020 01:00



Biology, 28.11.2020 01:00

Biology, 28.11.2020 01:00

Business, 28.11.2020 01:00


Mathematics, 28.11.2020 01:00

Mathematics, 28.11.2020 01:00

Mathematics, 28.11.2020 01:00

Chemistry, 28.11.2020 01:00

History, 28.11.2020 01:00

Geography, 28.11.2020 01:00




