
Chemistry, 08.04.2020 02:35 brianadee800
In the reaction of nitrogen gas, N2, with hydrogen gas, H2, to form ammonia gas, NH3, how many moles of hydrogen are needed to react with two moles of nitrogen? N2 + 3H2 → 2NH3 Select one: A. 5.710 moles B. 4 moles C. 2 moles D. 8 moles E. 6 moles

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 23.06.2019 00:00
Before it was launched, a helium-filled balloon had a pressure of 201 kpa at a temperature of 27°c. at an altitude of 15,000 m, the pressure had decreased to 2.5 kpa and the temperature had dropped to -14 °c. the volume of the balloon increased to 59.3 m3. what is the original volume of the balloon? 13 m3 0.85 m3 0.077 m3 1.17 m3
Answers: 3

Chemistry, 23.06.2019 00:20
What type of context clue you understand the meaning of quandary?
Answers: 3

Chemistry, 23.06.2019 05:30
The term gas is limited to those substances that exist in the gaseous state at
Answers: 1
You know the right answer?
In the reaction of nitrogen gas, N2, with hydrogen gas, H2, to form ammonia gas, NH3, how many moles...
Questions

Mathematics, 01.09.2019 05:00

Chemistry, 01.09.2019 05:00


Physics, 01.09.2019 05:00

Mathematics, 01.09.2019 05:00


Mathematics, 01.09.2019 05:00




Social Studies, 01.09.2019 05:00


Social Studies, 01.09.2019 05:00







Chemistry, 01.09.2019 05:00

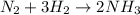
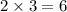 moles of hydrogen gas
moles of hydrogen gas

