
Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 02:30
98 ! and brainliest plz ! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1
You know the right answer?
How many grams of H20 gas is 3.5 L?...
Questions

Mathematics, 04.02.2021 04:50

Chemistry, 04.02.2021 04:50

Chemistry, 04.02.2021 04:50

History, 04.02.2021 04:50

Mathematics, 04.02.2021 04:50

English, 04.02.2021 04:50

Mathematics, 04.02.2021 04:50


Mathematics, 04.02.2021 04:50

Mathematics, 04.02.2021 04:50

Mathematics, 04.02.2021 04:50

Mathematics, 04.02.2021 04:50


Biology, 04.02.2021 04:50

Mathematics, 04.02.2021 04:50



Mathematics, 04.02.2021 04:50

Mathematics, 04.02.2021 04:50

Mathematics, 04.02.2021 05:00

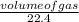
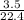 = 0.16mole
= 0.16mole

