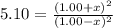Chemistry, 07.04.2020 21:10 kenishawilkinsoy4mgw
Carbon Monoxide reacts with steam to produce carbon dioxide and hydrogen. At 700K, the equilibrium constant is 5.10. Calculate the equilibrium concentrations of all species if 1.00mol of each component is mixed in a 1.00L flask.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Reaction rate depends on how many molecules are coming into contact with each other with enough energy to react. increasing the temperature of the reactants will increase -
Answers: 3

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3

Chemistry, 23.06.2019 01:30
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2
You know the right answer?
Carbon Monoxide reacts with steam to produce carbon dioxide and hydrogen. At 700K, the equilibrium c...
Questions


Mathematics, 23.11.2019 14:31





Mathematics, 23.11.2019 14:31

Mathematics, 23.11.2019 14:31

History, 23.11.2019 14:31


Chemistry, 23.11.2019 14:31

Mathematics, 23.11.2019 14:31

Mathematics, 23.11.2019 14:31

Mathematics, 23.11.2019 14:31



Computers and Technology, 23.11.2019 14:31



Mathematics, 23.11.2019 14:31

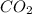 at equilibrium= 1.386 M
at equilibrium= 1.386 M 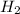 at equilibrium = 1.386 M
at equilibrium = 1.386 M 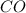 at equilibrium = 0.614 M
at equilibrium = 0.614 M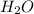 at equilibrium= 0.614 M
at equilibrium= 0.614 M 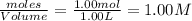
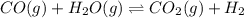
![K_c=\frac{[CO_2]\times [H_2]}{[CO]\times [H_2O]}](/tpl/images/0587/3047/a0f86.png)