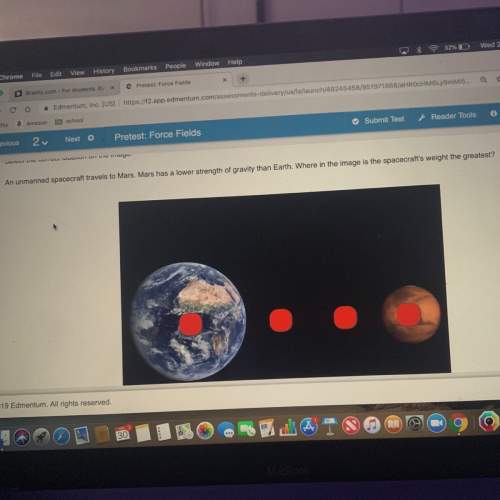
Which of the following statements is true? A. Lithium will react to give up an electron however that electron can only be used to make an anion of a metal B. Lithium is most easily oxidized of the metals listed on the activity series and therefore it will most easily give electrons to metal cations C. Lithium is least easily oxidized of the metals listed on the activity series and therefore it is least likely to give electrons to metal cations D. Lithium will only react with metal ions that require the addition of one electron because of the fact that lithium only gives up one electron

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 21:00
One similarity and one difference between an element and a mixture of elements
Answers: 1

Chemistry, 22.06.2019 22:30
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1

Chemistry, 23.06.2019 12:30
The equilibrium constant kc for the reaction 2 nocl(g) → 2 no(g) + cl2(g) is 0.453 at a certain temperature. a mixture of nocl, no, and cl2 with concentrations 1.30, 1.20, and 0.600 m, respectively, was introduced into a container at this temperature. which of the following is true? 1. no apparent reaction takes place. 2. [cl2] = 0.30 m at equilibrium. 3. nocl(g) is produced until equilibrium is reached. 4. [nocl] = [no] = [cl2] at equilibrium. 5. cl2(g) is produced until equilibrium is
Answers: 3
You know the right answer?
Which of the following statements is true? A. Lithium will react to give up an electron however that...
Questions

Social Studies, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Spanish, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

English, 13.09.2020 16:01

Chemistry, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Biology, 13.09.2020 16:01

History, 13.09.2020 16:01

Business, 13.09.2020 16:01

English, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

English, 13.09.2020 16:01

Mathematics, 13.09.2020 17:01

Chemistry, 13.09.2020 17:01

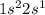 . It will attain stability by losing an electron.
. It will attain stability by losing an electron.