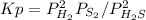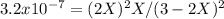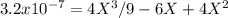Chemistry, 07.04.2020 19:18 litttyyyu33411
At 800 K, the equilibrium constant, Kp, for the following reaction is 3.2 × 10–7. 2 H2S(g) ⇌ 2 H2(g) + S2(g) A reaction vessel at 800 K initially contains 3.00 atm of H2S. If the reaction is allowed to equilibrate, what is the equilibrium pressure of S2?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Pbco3 –> pbo+ co2. how many liters of carbon dioxide gas is produced from the decomposition of 32 grams of lead (ll) carbonate?
Answers: 1

Chemistry, 22.06.2019 12:00
What is the subscript for oxygen in its molecular formula
Answers: 1

Chemistry, 22.06.2019 19:30
What is the area in square meters of 448 g ai foil that has a thickness of 23921 nm? the density is 2.70 g/cm
Answers: 3

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
At 800 K, the equilibrium constant, Kp, for the following reaction is 3.2 × 10–7. 2 H2S(g) ⇌ 2 H2(g)...
Questions

Biology, 09.07.2019 00:30

Physics, 09.07.2019 00:30

Mathematics, 09.07.2019 00:30

Social Studies, 09.07.2019 00:30

Mathematics, 09.07.2019 00:30

Mathematics, 09.07.2019 00:30

Mathematics, 09.07.2019 00:30

Mathematics, 09.07.2019 00:30

Biology, 09.07.2019 00:30

English, 09.07.2019 00:30



Mathematics, 09.07.2019 00:30

Mathematics, 09.07.2019 00:30



Advanced Placement (AP), 09.07.2019 00:30

Mathematics, 09.07.2019 00:30