
Chemistry, 07.04.2020 17:03 ashley110608
Reaction A has a high activation energy, whereas reacton B has a low activation energy. Which of the statements about reaction A and reaction B are true? Reaction B is likely to occur at a faster rate than reaction A. Reaction A is more likely to occur at all than reaction B. Reaction B is more likely to occur at all than reaction A. Reaction A is likely to occur at a faster rate than reaction B.

Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 19:40
What causes different colors to appear in the sky? the absorption of light by air molecules the reflection of light by bodies of water the greenhouse effect in earth's atmosphere the scattering and reflection of light by dust particles
Answers: 2
You know the right answer?
Reaction A has a high activation energy, whereas reacton B has a low activation energy. Which of the...
Questions


English, 06.07.2020 08:01

Mathematics, 06.07.2020 08:01

Mathematics, 06.07.2020 08:01


Mathematics, 06.07.2020 08:01





Mathematics, 06.07.2020 09:01


Computers and Technology, 06.07.2020 09:01

Mathematics, 06.07.2020 09:01

Biology, 06.07.2020 09:01

Business, 06.07.2020 09:01



Mathematics, 06.07.2020 09:01

Mathematics, 06.07.2020 09:01

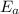 , and it is measured in Joules (J), KiloJoules (KJ) or Kilocalories per mole (Kcal/mol)
, and it is measured in Joules (J), KiloJoules (KJ) or Kilocalories per mole (Kcal/mol)

