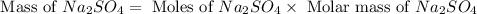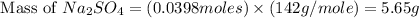Chemistry, 06.04.2020 16:59 zitterkoph
Aqueous sulfuric acid (H2SO4) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium sulfate (Na2SO4) and liquid water (H2O). What is the theoretical yield of sodium sulfate formed from the reaction of 3.9 g of sulfuric acid and 1.8g of sodium hydroxide?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
50 pts plz what is the physical state of matter of baking soda.
Answers: 1

Chemistry, 22.06.2019 08:30
What is the independent variable in this investigation? mass volume sample number substance density
Answers: 3

Chemistry, 22.06.2019 21:50
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state.a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2

Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1
You know the right answer?
Aqueous sulfuric acid (H2SO4) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium su...
Questions

Mathematics, 07.10.2021 08:20

Mathematics, 07.10.2021 08:20



Social Studies, 07.10.2021 08:20



Spanish, 07.10.2021 08:20


Mathematics, 07.10.2021 08:20

Computers and Technology, 07.10.2021 08:20

Chemistry, 07.10.2021 08:20

Mathematics, 07.10.2021 08:30

Biology, 07.10.2021 08:30


Mathematics, 07.10.2021 08:30

Arts, 07.10.2021 08:30

Biology, 07.10.2021 08:30

Biology, 07.10.2021 08:30

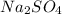 is, 5.65 grams.
is, 5.65 grams.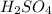 = 3.9 g
= 3.9 g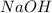 = 7.8 g
= 7.8 g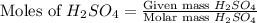
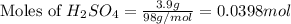
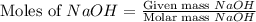
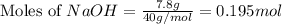
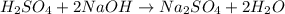
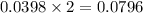 moles of
moles of