Chemistry, 04.04.2020 14:34 wirchakethan23
The reform reaction between steam and gaseous methane () produces "synthesis gas," a mixture of carbon monoxide gas and dihydrogen gas. Synthesis gas is one of the most widely used industrial chemicals, and is the major industrial source of hydrogen. Suppose a chemical engineer studying a new catalyst for the reform reaction finds that liters per second of methane are consumed when the reaction is run at and . Calculate the rate at which dihydrogen is being produced. Give your answer in kilograms per second. Round your answer to significant digits..

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3

Chemistry, 22.06.2019 08:30
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 14:00
What term describes technology that operates on an atomic level
Answers: 2

Chemistry, 22.06.2019 16:30
Correct relationship between molecular formula and empirical formula
Answers: 1
You know the right answer?
The reform reaction between steam and gaseous methane () produces "synthesis gas," a mixture of carb...
Questions









Mathematics, 25.06.2019 17:30

Advanced Placement (AP), 25.06.2019 17:30



Biology, 25.06.2019 17:30

History, 25.06.2019 17:30



Chemistry, 25.06.2019 17:30


Mathematics, 25.06.2019 17:30

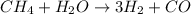
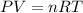
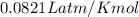
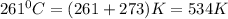
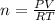
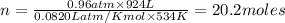
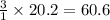 moles of hydrogen
moles of hydrogen