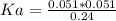Calculate the Ka for the following acid. Determine if it is a strong or weak acid. HClO2(aq) dissolves in aqueous solution to form H+(aq) and ClO2−(aq). At equilibrium, the concentrations of each of the species are as follows: [HClO2]=0.24M [H+]=0.051M [ClO2−]=0.051M

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no]2[o2]
Answers: 3

Chemistry, 22.06.2019 03:00
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3

Chemistry, 22.06.2019 21:30
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3

Chemistry, 22.06.2019 23:30
If it is an isoelectronic series select true, if not select false. o2-, s2-, se2-, te2- na+, k+, rb+, cs+ n3-, p3-, as3-, sb3- ag, cd+, sn3+, sb4+ f-, cl-, br-, i- f-, ne, na+, mg2+ s2-, s, s6+
Answers: 1
You know the right answer?
Calculate the Ka for the following acid. Determine if it is a strong or weak acid. HClO2(aq) dissolv...
Questions




Mathematics, 29.03.2021 21:20


Mathematics, 29.03.2021 21:20


Mathematics, 29.03.2021 21:20


Mathematics, 29.03.2021 21:20

Mathematics, 29.03.2021 21:20


History, 29.03.2021 21:20


English, 29.03.2021 21:20

Health, 29.03.2021 21:20

Social Studies, 29.03.2021 21:20

Mathematics, 29.03.2021 21:20


Chemistry, 29.03.2021 21:20


![[HClO_2]=0.24M](/tpl/images/0582/3449/64bd9.png)
![[H^+]=0.051M](/tpl/images/0582/3449/e1645.png)
![[ClO_2^-]=0.051M](/tpl/images/0582/3449/4a4bd.png)
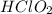 is
is
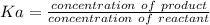
![= \frac{[H^+][ClO_2^-]}{[HClO_2]}](/tpl/images/0582/3449/87292.png)