
Chemistry, 04.04.2020 12:08 ErrorNameTaken505
Lead (II) sulfide, PbS, reacts with oxygen gas to produce lead (II) oxide and sulfur dioxide. If 0.750 moles of O2 were used during this chemical reaction, how many grams of lead (II) oxide would be produced?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:50
2. you__turn left on a red light if you are in the left-most lane of a one-way street, you're turning into the left-most lane of a one-way street, and no nearby sign prohibits the turn.
Answers: 2

Chemistry, 22.06.2019 06:00
Oxidation-reduction reactions (often called "redox" for short) are reactions that involve the transfer of electrons from one species to another. oxidation states, or oxidation numbers, allow chemists to keep track of these electron transfers. in general, one element will lose electrons (oxidation), with the result that it will increase in oxidation number, and another element will gain electrons (reduction), thereby decreasing in oxidation number. the species that is oxidized is called the reducing agent or reductant. the species that is reduced is called the oxidizing agent or oxidant. to sum up: oxidation = increase in oxidation state = loss of electrons = reducing agent reduction = decrease in oxidation state = gain of electrons = oxidizing agent part a which element is oxidized in this reaction? fe2o3+3co→2fe+3co2 enter the elemental symbol. view available hint(s) is oxidized part b which element is reduced in this reaction? 2hcl+2kmno4+3h2c2o4→6co2+2mno2+2kcl+4h2o enter the elemental symbol. view available hint(s) is reduced
Answers: 1

Chemistry, 22.06.2019 08:00
This classification of drug typically changes the brain's chemistry and reduces its ability to create its own endorphins.
Answers: 1

Chemistry, 22.06.2019 20:30
Which states of matter have particles that move independently of one another with very little attraction?
Answers: 1
You know the right answer?
Lead (II) sulfide, PbS, reacts with oxygen gas to produce lead (II) oxide and sulfur dioxide. If 0.7...
Questions


Social Studies, 03.01.2022 01:00


Mathematics, 03.01.2022 01:00

Social Studies, 03.01.2022 01:00

English, 03.01.2022 01:00

SAT, 03.01.2022 01:00

Mathematics, 03.01.2022 01:00

Chemistry, 03.01.2022 01:00

Mathematics, 03.01.2022 01:00

Mathematics, 03.01.2022 01:00

English, 03.01.2022 01:00

Mathematics, 03.01.2022 01:00


History, 03.01.2022 01:00



Mathematics, 03.01.2022 01:00

Mathematics, 03.01.2022 01:00

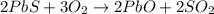
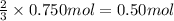 of lead (II) oxide
of lead (II) oxide


