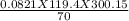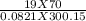A 70-liter tank of oxygen gas at 27 degree Celcius and 42.0 atm springs a leak overnight. when the tank was found in the morning, the pressure in the tank had dropped to 19.0 atm. If the tank originally held 119.4 moles of oxygen gas, how many moles of gas were left the next morning, assuming the temperature and volume of the rank stayed constant?.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 08:30
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

You know the right answer?
A 70-liter tank of oxygen gas at 27 degree Celcius and 42.0 atm springs a leak overnight. when the t...
Questions



Mathematics, 05.05.2021 22:50





Mathematics, 05.05.2021 22:50

Mathematics, 05.05.2021 22:50

English, 05.05.2021 22:50

Mathematics, 05.05.2021 22:50



Mathematics, 05.05.2021 22:50

Physics, 05.05.2021 22:50

Arts, 05.05.2021 22:50

Mathematics, 05.05.2021 22:50

Physics, 05.05.2021 22:50

Mathematics, 05.05.2021 22:50