
Chemistry, 04.04.2020 11:01 deebroussard
Find the pH during the titration of 20.00 mL of 0.1000 M butanoic acid, CH3CH2CH2COOH (Ka = 1.54 x 10^-5), with 0.1000 M NaOH solution after the following additions of titrant.
(a) 10.00 mL: pH =
(b) 20.10 mL: pH =
(c) 25.00 mL: pH =

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
18. use the activity series to predict whether the following synthesis reaction will occur. write the chemical equations for the reaction if it's predicted to occur. (s) + o2(g) -> *note: it is possible.*
Answers: 1

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 04:30
How much energy is made when a pice of wood burns. how do you know
Answers: 2

Chemistry, 22.06.2019 14:00
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2
You know the right answer?
Find the pH during the titration of 20.00 mL of 0.1000 M butanoic acid, CH3CH2CH2COOH (Ka = 1.54 x 1...
Questions


English, 05.01.2021 17:10



Mathematics, 05.01.2021 17:10


Mathematics, 05.01.2021 17:10


Health, 05.01.2021 17:10


Mathematics, 05.01.2021 17:10



English, 05.01.2021 17:10


Physics, 05.01.2021 17:10

Mathematics, 05.01.2021 17:10

Mathematics, 05.01.2021 17:10


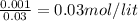
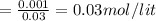
![pH=pK_{a}+log\frac{[conjugate base]}{[acid]} \\\\=-log(1.54X10^{-5} )+log\frac{0.03}{0.03} \\\\=4.81](/tpl/images/0582/2071/3377e.png)




