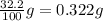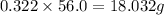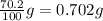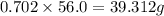Chemistry, 04.04.2020 10:59 iklassibrahim123
Consider the solubilities of a particular solute at two different temperatures. Temperature ( ∘ C ) Solubility ( g / 100 g H 2 O ) 20.0 32.2 30.0 70.2 Suppose a saturated solution of this solute was made using 56.0 g H 2 O at 20.0 °C. How much more solute can be added if the temperature is increased to 30.0 ∘ C? mass:

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 10:30
Aglow stick contains a glass vial with chemicals. when the glow stick is bent, the vial breaks and the chemicals react to produce a glow. a science student observes that a glow stick kept in the freezer glows for a longer duration than a glow stick kept at room temperature. what conclusion can be drawn based on the observation? be sure to note the outcome and test variables in the conclusion.
Answers: 1

Chemistry, 22.06.2019 22:00
Ill give u brainliest pls how is mass of carbon conserved during cellular respiration
Answers: 1

Chemistry, 22.06.2019 23:30
With the largest atoms and the smallest number of valence electrons and with the smallest atoms and the greatest number of valence electrons are the most reactive. a. nonmetals; metals b. nonmetals; transition elements c. transition elements; metals d. metals; nonmetals
Answers: 3
You know the right answer?
Consider the solubilities of a particular solute at two different temperatures. Temperature ( ∘ C )...
Questions

History, 01.04.2021 07:20

SAT, 01.04.2021 07:20

Mathematics, 01.04.2021 07:20

Mathematics, 01.04.2021 07:20


Mathematics, 01.04.2021 07:20

English, 01.04.2021 07:20

Mathematics, 01.04.2021 07:20

Mathematics, 01.04.2021 07:20





Chemistry, 01.04.2021 07:20


Mathematics, 01.04.2021 07:20


Mathematics, 01.04.2021 07:20

Mathematics, 01.04.2021 07:20

Physics, 01.04.2021 07:20