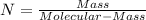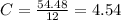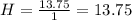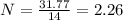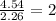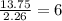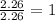Chemistry, 03.04.2020 03:29 danding1593
What is the empirical formula for a compound that is 54.48% carbon, 13.75% hydrogen, and 31.77 % nitrogen?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 16:00
How could a student test the effect of removing heat from a gas that is stored in a sealed container? what must occur in order for matter to change states?
Answers: 2

Chemistry, 22.06.2019 17:00
How can a give a full method for the experiment of separating sand from water by filtration? 1-materials 2-steps 3-conclusion also for water and salt separated by the evaporation or distillation process
Answers: 1
You know the right answer?
What is the empirical formula for a compound that is 54.48% carbon, 13.75% hydrogen, and 31.77 % nit...
Questions

Mathematics, 11.11.2020 01:00


Health, 11.11.2020 01:00

English, 11.11.2020 01:00

Mathematics, 11.11.2020 01:00



Mathematics, 11.11.2020 01:00

Chemistry, 11.11.2020 01:00

Mathematics, 11.11.2020 01:00

Arts, 11.11.2020 01:00

Mathematics, 11.11.2020 01:00

Geography, 11.11.2020 01:00

Mathematics, 11.11.2020 01:00


Advanced Placement (AP), 11.11.2020 01:00

Mathematics, 11.11.2020 01:00



English, 11.11.2020 01:00

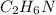 .
.