Chemistry, 02.04.2020 18:45 wendymtz2004
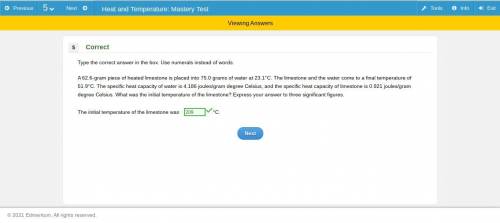
A 62.6-gram piece of heated limestone is placed into 75.0 grams of water at 23.1°C. The limestone and the water come to a final temperature of 51.9°C. The specific heat capacity of water is 4.186 joules/gram degree Celsius, and the specific heat capacity of limestone is 0.921 joules/gram degree Celsius. What was the initial temperature of the limestone? Express your answer to three significant figures

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 11:50
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be
Answers: 1

You know the right answer?
A 62.6-gram piece of heated limestone is placed into 75.0 grams of water at 23.1°C. The limestone an...
Questions





Mathematics, 24.11.2021 09:30

English, 24.11.2021 09:30

Social Studies, 24.11.2021 09:30





Mathematics, 24.11.2021 09:30



Computers and Technology, 24.11.2021 09:30

Mathematics, 24.11.2021 09:30

Mathematics, 24.11.2021 09:30

Biology, 24.11.2021 09:30


Mathematics, 24.11.2021 09:30