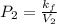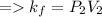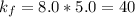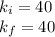Chemistry, 02.04.2020 02:35 LadyHolmes67
A certain gas is present in a 10.0 L cylinder at 4.0 atm pressure. If the pressure is increased to 8.0 atm the volume of the gas decreases to 5.0 L . Find the two constants ki, the initial value of k, and kf, the final value of k, to verify whether the gas obeys Boyle’s law by entering the numerical values for ki and kf in the space provided.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Which two scientific disciplines are bridged by pharmaceutical drugs? chemistry and forensics chemistry and medicine biology and forensics biology and criminology
Answers: 3

Chemistry, 22.06.2019 12:20
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1


Chemistry, 22.06.2019 14:30
Which of the following is not one of the steps in the scientific method a. hypothesize b. summarize c. analyze d. familiarize
Answers: 3
You know the right answer?
A certain gas is present in a 10.0 L cylinder at 4.0 atm pressure. If the pressure is increased to 8...
Questions


Mathematics, 08.01.2021 02:20


Mathematics, 08.01.2021 02:20



Mathematics, 08.01.2021 02:20

Mathematics, 08.01.2021 02:20

Biology, 08.01.2021 02:20




Mathematics, 08.01.2021 02:20




History, 08.01.2021 02:20



Physics, 08.01.2021 02:20

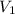 = 12.0 L
= 12.0 L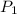 = 4.0 atm
= 4.0 atm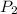 = 8.0 atm
= 8.0 atm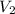 = 6.0 L
= 6.0 L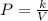
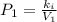
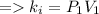
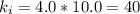
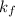 = ?
= ?