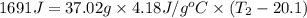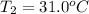Chemistry, 02.04.2020 01:26 kevinkingpin
What is the final temperature of the solution formed when 1.52 g of NaOH is added to 35.5 g of water at 20.1 °C in a calorimeter? NaOH (s) → Na+ (aq) + OH– (aq) ∆H = -44.5 kJ/mol

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 08:30
In the millikan oil drop experiment they determined that every drop had a charge which was a while number multiple of -1.60x10^-19. if a drop has a total charge of -9.60x10^-19 then how many excess electrons are contained within the drop?
Answers: 2

Chemistry, 22.06.2019 11:00
When hydrochloric acid reacts with potassium hydroxide solution, the following reaction occurs. hcl (aq) + koh (aq) h2o (l) + kcl (aq) the reaction gives off heat energy, so it is an reaction.
Answers: 1

Chemistry, 22.06.2019 22:00
11) burning your hand when accidentally touching a hot plate is an example of which heat transfer? a. conduction b. convection c. radiation d. none of these
Answers: 2
You know the right answer?
What is the final temperature of the solution formed when 1.52 g of NaOH is added to 35.5 g of water...
Questions



Biology, 05.10.2019 15:30


History, 05.10.2019 15:30

Business, 05.10.2019 15:30

History, 05.10.2019 15:30






Spanish, 05.10.2019 15:30


Mathematics, 05.10.2019 15:30

History, 05.10.2019 15:30


History, 05.10.2019 15:30


Physics, 05.10.2019 15:30

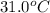
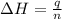
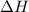 = enthalpy change = -44.5 kJ/mol
= enthalpy change = -44.5 kJ/mol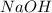 = 1.52 g
= 1.52 g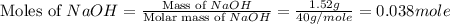
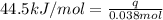
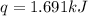
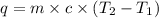
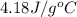
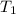 = initial temperature =
= initial temperature = 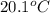
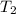 = final temperature = ?
= final temperature = ?