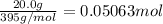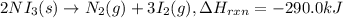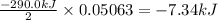Chemistry, 02.04.2020 01:13 wichserc3399
The decomposition of NI3 to form N2 and I2 releases −290.0 kJ of energy. The reaction can be represented as 2NI3(s)→N2(g)+3I2(g), ΔHrxn=−290.0 kJ Find the change in enthaply when 20.0 g of NI3 decomposes.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:20
Concerning the 10.0 ml of 0.50 m nacl to 100 ml of solution: when a solution is diluted, does it change the number of moles dissolved?
Answers: 3

Chemistry, 22.06.2019 08:30
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3


Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3
You know the right answer?
The decomposition of NI3 to form N2 and I2 releases −290.0 kJ of energy. The reaction can be represe...
Questions


Mathematics, 26.07.2019 16:00




Business, 26.07.2019 16:00







Mathematics, 26.07.2019 16:00

Chemistry, 26.07.2019 16:00


Biology, 26.07.2019 16:00



Mathematics, 26.07.2019 16:00

Mathematics, 26.07.2019 16:00