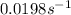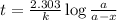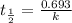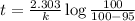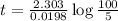Chemistry, 02.04.2020 03:27 cuddleslugsovifat
Be sure to answer all parts. The thermal decomposition of phosphine (PH3) into phosphorus and molecular hydrogen is a first-order reaction: 4PH3(g) → P4(g) + 6H2(g) The half-life of the reaction is 35.0 s at 680°C.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:40
Base your answer on the information below and on your knowledge of chemistry. nitrogen dioxide, no2, is a dark brown gas that is used to make nitric acid and to bleach flour. nitrogen dioxide has a boiling point of 294 k at 101.3 kpa. in a rigid cylinder with a movable piston, nitrogen dioxide can be in equilibrium with colorless dinitrogen tetroxide, n2o4. this equilibrium is represented by the equation below. 2no2(g) n2o4(g) + 58kj at standard pressure, compare the strength of intermolecular forces in no2(g) to the strength of intermolecular forces in n2(g).
Answers: 2

Chemistry, 22.06.2019 02:40
How many liters of hydrogen gas will be produced at stp from the reaction of 7.179×10^23 atoms of magnesium with 54.219g of phosphoric acid (h3po4) the equation is 3mg + 2h3(> mg(po4)2+3h2
Answers: 1

Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3
You know the right answer?
Be sure to answer all parts. The thermal decomposition of phosphine (PH3) into phosphorus and molecu...
Questions

Mathematics, 10.04.2021 19:40




Mathematics, 10.04.2021 19:50

Computers and Technology, 10.04.2021 19:50



Geography, 10.04.2021 19:50

Mathematics, 10.04.2021 19:50



English, 10.04.2021 19:50


Mathematics, 10.04.2021 19:50

History, 10.04.2021 19:50


Physics, 10.04.2021 19:50

Mathematics, 10.04.2021 19:50

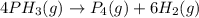 The half-life of the reaction is 35.0 s at 680°C. Calculate the first order rate constant.
The half-life of the reaction is 35.0 s at 680°C. Calculate the first order rate constant.