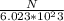Chemistry, 01.04.2020 12:54 alanis337455p4xzek
A sample of calcium phosphate was found to have a mass of 125.3 g. How many molecules were contained in the sample?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1

Chemistry, 22.06.2019 19:50
If a gas has an initial pressure of 101kpa and a volume of 10l, then it expands to a volume of 20l, what is the new pressure?
Answers: 2

Chemistry, 22.06.2019 21:50
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state.a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2

Chemistry, 23.06.2019 00:50
50 points! need answer asap. what type of organic compound contains the following functional group? (2 points)
Answers: 3
You know the right answer?
A sample of calcium phosphate was found to have a mass of 125.3 g. How many molecules were contained...
Questions



Mathematics, 24.07.2019 17:00


Mathematics, 24.07.2019 17:00

Mathematics, 24.07.2019 17:00

Mathematics, 24.07.2019 17:00

History, 24.07.2019 17:00



Social Studies, 24.07.2019 17:00


Biology, 24.07.2019 17:00



Biology, 24.07.2019 17:00




History, 24.07.2019 17:00

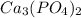 ) = 125.3 grams
) = 125.3 grams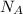 ) = 6.023 × 10^23 molecules
) = 6.023 × 10^23 molecules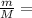 N÷
N÷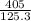 =
=