
Chemistry, 01.04.2020 05:43 payshencec21
For the equilibrium
2IBr(g) ⇌ I₂(g) + Br₂(g) Kc = 8.50 × 10⁻³ at 150°C.
If 0.0600 mol of IBr is placed in a 1.0-L container, what is the partial pressure of I₂(g) in atm after equilibrium is reached?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 20:00
Listenbase your answer to the question on the information below.nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body.cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment.which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2



Chemistry, 23.06.2019 11:00
Just on number 2 (all parts), and if you do answer explain in detail
Answers: 3
You know the right answer?
For the equilibrium
2IBr(g) ⇌ I₂(g) + Br₂(g) Kc = 8.50 × 10⁻³ at 150°C.
If 0...
2IBr(g) ⇌ I₂(g) + Br₂(g) Kc = 8.50 × 10⁻³ at 150°C.
If 0...
Questions



Mathematics, 30.10.2020 22:30


English, 30.10.2020 22:40

Mathematics, 30.10.2020 22:40



History, 30.10.2020 22:40

Mathematics, 30.10.2020 22:40

Health, 30.10.2020 22:40



Mathematics, 30.10.2020 22:40

Health, 30.10.2020 22:40


Mathematics, 30.10.2020 22:40


Mathematics, 30.10.2020 22:40

Mathematics, 30.10.2020 22:40

![[IBr]i = \frac{0.0600mol}{1.0L} = 0.060M](/tpl/images/0575/0702/18a8d.png)
![Kc = 8.50 \times 10^{-3} = \frac{[I_2][Br_2]}{[IBr]^{2} } = \frac{x^{2} }{(0.060-x)^{2} } \\\\x = 0.00507](/tpl/images/0575/0702/c2c7d.png)
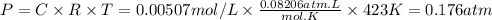
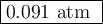
![\text{[IBr]} = \dfrac{\text{0.0600 mol}}{\text{1.0 L}} = \text{0.0600 mol/L}](/tpl/images/0575/0702/bae1e.png)
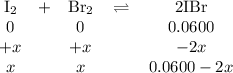
![\begin{array}{rcl}K_{\text{c}}&=&\dfrac{\text{[IBr]}^{2}} {\text{[I$_{2}$][Br]$_{2}$}}\\\\8.50 \times 10^{-2}&=&{\dfrac{(0.0600 - 2x)^{2}}{x^{2}}}& &\\\\0.2915x & = &{\dfrac{0.0600 - 2x}{x}}& &\\\\0.2915x & = &0.0600 - 2x\\\\2.2915x & = & 0.0600\\x & = & \textbf{0.026 18 mol/L}\\\end{array}\\](/tpl/images/0575/0702/36fc0.png)



