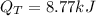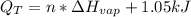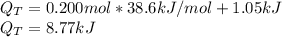Ethanol boils at 78.4 °C with \DeltaΔHvap = 38.6 kJ/mol. A 0.200-mol sample of ethanol is heated from some colder temperature up to 78.4 °C, which requires 1.05 kJ of heat, and then vaporized. What will be the total amount of heat required (for both the heating and the vaporizing)?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:00
3. if the dartboard below is used to model an atom, which subatomic particles would be located at z?
Answers: 2

Chemistry, 21.06.2019 20:20
Concerning the 10.0 ml of 0.50 m nacl to 100 ml of solution: when a solution is diluted, does it change the number of moles dissolved?
Answers: 3

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1
You know the right answer?
Ethanol boils at 78.4 °C with \DeltaΔHvap = 38.6 kJ/mol. A 0.200-mol sample of ethanol is heated fro...
Questions


Mathematics, 31.08.2019 06:00


Physics, 31.08.2019 06:00

Chemistry, 31.08.2019 06:00



Chemistry, 31.08.2019 06:00





English, 31.08.2019 06:00

Mathematics, 31.08.2019 06:00

English, 31.08.2019 06:00

Biology, 31.08.2019 06:00

Chemistry, 31.08.2019 06:00

Social Studies, 31.08.2019 06:00


Mathematics, 31.08.2019 06:00