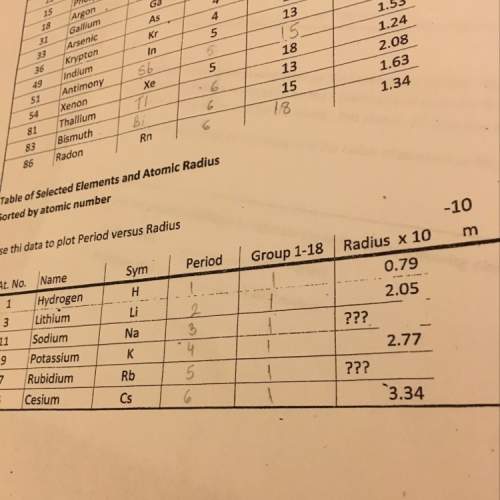
Chemistry, 31.03.2020 01:57 pastmarion
In the activity, click on the E∘cellEcell∘ and KeqKeq quantities to observe how they are related. Use this relation to calculate KeqKeq for the following redox reaction that occurs in an electrochemical cell having two electrodes: a cathode and an anode. The two half-reactions that occur in the cell are Cu2+(aq)+2e−→Cu(s)Cu2+(aq)+2e−→Cu(s ) and Fe(s)→Fe2+(aq)+2e−

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:00
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1


Chemistry, 23.06.2019 09:30
Sheela and her brother hari were sitting in the living room, watching tv. suddenly hari said that he thinks something is burning in the other room. how did he get the burning smell?
Answers: 3

Chemistry, 23.06.2019 11:30
How do you calculate the mass of a product when the amounts of more than one reactant are given?
Answers: 3
You know the right answer?
In the activity, click on the E∘cellEcell∘ and KeqKeq quantities to observe how they are related. Us...
Questions


Mathematics, 07.04.2021 22:50


Mathematics, 07.04.2021 22:50

Physics, 07.04.2021 22:50



Computers and Technology, 07.04.2021 22:50

Mathematics, 07.04.2021 22:50


Mathematics, 07.04.2021 22:50



Computers and Technology, 07.04.2021 22:50


Mathematics, 07.04.2021 22:50


English, 07.04.2021 22:50


Mathematics, 07.04.2021 22:50