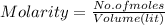Chemistry, 31.03.2020 01:28 rainbow7020
A sample of 8.4 grams of NaOH is dissolved into 620 mL of aqueous 0.250 M NaOH (assume no volume change). This solution is then poured into 1.65 gallons of water. (You may assume that the two volumes can be added.) What is the concentration of NaOH in the final solution

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:40
Asingle atom of an element has 21 neutrons, 20 electrons, and 20 protons. which element is it? ok o z
Answers: 1

Chemistry, 21.06.2019 20:30
1. calculate the approximate enthalpy of the reaction in joules. estimate that 1.0 ml of vinegar has the same thermal mass as 1.0 ml of water. iqnore the thermal mass of th sodium bicarbonate. note: it takes about 4.2 joules () to change 1.0 gram (1.0ml) of water 1.0 c
Answers: 2

Chemistry, 22.06.2019 14:30
According to le chatelier’s principle, a system in chemical equilibrium responds to stress by shifting the equilibrium in a direction that reduces the stress. normalizes the stress. increases the stress. changes the stress.
Answers: 1

Chemistry, 22.06.2019 18:30
The table lists the lattice energies of some compounds.compoundlattice energy (kj/mol)lif –1,036licl –853naf –923kf –821nacl –786which statement about crystal lattice energy is best supported by the information in the table? the lattice energy increases as cations get smaller, as shown by lif and kf.the lattice energy increases as the cations get larger, as shown by lif and licl.the lattice energy decreases as cations get smaller, as shown by nacl and naf.the lattice energy decreases as the cations get smaller, as shown by naf and kf.
Answers: 3
You know the right answer?
A sample of 8.4 grams of NaOH is dissolved into 620 mL of aqueous 0.250 M NaOH (assume no volume cha...
Questions


Mathematics, 18.01.2021 01:00

Mathematics, 18.01.2021 01:00


Social Studies, 18.01.2021 01:00



Mathematics, 18.01.2021 01:00

English, 18.01.2021 01:00





Mathematics, 18.01.2021 01:00

Mathematics, 18.01.2021 01:00

Mathematics, 18.01.2021 01:00

Mathematics, 18.01.2021 01:00



Mathematics, 18.01.2021 01:00

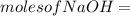
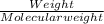
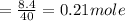 620 ml of aqueous 0.25 M NaOH
620 ml of aqueous 0.25 M NaOH 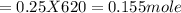 Total moles = 0.21 + 0.155 = 0.365 moles1.65 gallons = 3.785 lit x 1.65 = 6.245929 lit (∵ 1 gallon = 3.785 lit)
Total moles = 0.21 + 0.155 = 0.365 moles1.65 gallons = 3.785 lit x 1.65 = 6.245929 lit (∵ 1 gallon = 3.785 lit)