
Chemistry, 30.03.2020 21:09 rvkanneh1713
(4) After addition of hydrochloric acid, a student removes the lower aqueous layer, extracts the aqueous phase with diethyl ether (0.5 mL), dries the diethyl ether layer with sodium sulfate and pours the ~0.5 mL of diethyl ether into an Erlenmeyer flask, rinsing the drying agent with more diethyl ether and pouring into the flask. Upon evaporation of the diethyl ether, the student is surprised to see no crystals present to recrystallize. Assuming Parts 1–3 were completed successfully, explain where the student lost their product.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Over the last 90 years, scientists have added to the body of evidence supporting the big bang theory. what is the latest piece of evidence discovered in 2014?
Answers: 1

Chemistry, 22.06.2019 11:00
Imagine that twenty i.u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2

Chemistry, 22.06.2019 22:30
Which statement best summarizes the importance of ernest rutherford’s gold foil experiment? it proved that all of john dalton’s postulates were true. it verified j. j. thomson’s work on the atomic structure. it showed that an electron circles a nucleus in a fixed-energy orbit. it showed that a nucleus occupies a small part of the whole atom.
Answers: 1

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
(4) After addition of hydrochloric acid, a student removes the lower aqueous layer, extracts the aqu...
Questions

Mathematics, 10.12.2020 18:10


English, 10.12.2020 18:10

Mathematics, 10.12.2020 18:10

History, 10.12.2020 18:10


Mathematics, 10.12.2020 18:10

Mathematics, 10.12.2020 18:10



Mathematics, 10.12.2020 18:10


Physics, 10.12.2020 18:10



Spanish, 10.12.2020 18:10

Mathematics, 10.12.2020 18:10


Mathematics, 10.12.2020 18:20

Mathematics, 10.12.2020 18:20

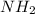 in the compound which have a tendency of forming
in the compound which have a tendency of forming 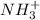 salt.
salt.

