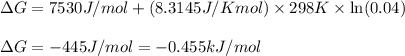Chemistry, 30.03.2020 21:38 zhuotingwu147
For the aqueous reaction dihydroxyacetone phosphate is the reactant and glyceraldehyde 3 phosphate is the product. dihydroxyacetone phosphate − ⇀ ↽ − glyceraldehyde − 3 − phosphate the standard change in Gibbs free energy is Δ G ° ' = 7.53 kJ/mol . Calculate Δ G for this reaction at 298 K when [dihydroxyacetone phosphate] = 0.100 M and [glyceraldehyde-3-phosphate] = 0.00400 M . Δ G = kJ / mol

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 00:30
Maria wants to determine which type of disinfectant kills the most bacteria. which of the following is the best way for maria to determine this? a. ask ten different companies that make disinfectants which type is best. b. put the same amount and species of bacteria on ten identical plates, and add ten different kinds of disinfectant to each plate. c. interview ten different people to determine which type of disinfectant they prefer. d. put the same amount and species of bacteria on ten identical plates, and add a different disinfectant to each plate.
Answers: 1

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2
You know the right answer?
For the aqueous reaction dihydroxyacetone phosphate is the reactant and glyceraldehyde 3 phosphate i...
Questions

Computers and Technology, 16.08.2021 18:00


English, 16.08.2021 18:00




History, 16.08.2021 18:00



Mathematics, 16.08.2021 18:00





Mathematics, 16.08.2021 18:00

Mathematics, 16.08.2021 18:10

Social Studies, 16.08.2021 18:10



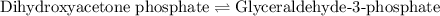
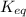 of above equation is:
of above equation is:![K_{eq}=\frac{\text{[Glyceraldehyde-3-phosphate]}}{\text{[Dihydroxyacetone phosphate]}}](/tpl/images/0571/3632/f265a.png)
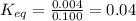
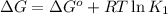
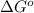 = Standard Gibbs free energy = 7.53 kJ/mol = 7530 J/mol
= Standard Gibbs free energy = 7.53 kJ/mol = 7530 J/mol