Chemistry, 30.03.2020 20:26 burnsmykala23
S) Suppose we now collect hydrogen gas, H2(g), over water at 21◦C in a vessel with total pressure of 743 Torr. If the hydrogen gas is produced by the reaction of aluminum with hydrochloric acid: 2Al(s) + 6HCl(aq) → 2AlCl3(aq) + 3H2(g) what volume of hydr

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:50
Suppose you got a low yield of benzoin from your benzoin condensation reaction and thus only have 0.300 g of benzoin to use as the starting material for this reaction. how much concentrated nitric acid should you add? (concentrated nitric acid is 15.8 m). write your answer in the form x.xx ml
Answers: 1

Chemistry, 22.06.2019 04:30
The big bang nucleosynthesis theory states that elements were produced in the first few minutes of the big bang while elements have their origins in the interiors of stars, forming much later in the history of the universe.
Answers: 1

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

You know the right answer?
S) Suppose we now collect hydrogen gas, H2(g), over water at 21◦C in a vessel with total pressure of...
Questions


English, 04.07.2019 07:00



History, 04.07.2019 07:00

Mathematics, 04.07.2019 07:00

Mathematics, 04.07.2019 07:00

Mathematics, 04.07.2019 07:00

History, 04.07.2019 07:00

Mathematics, 04.07.2019 07:00

Mathematics, 04.07.2019 07:00


Physics, 04.07.2019 07:00


Mathematics, 04.07.2019 07:00

History, 04.07.2019 07:00

Mathematics, 04.07.2019 07:00


Mathematics, 04.07.2019 07:00

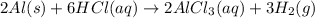
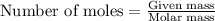
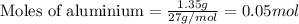
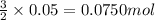 of hydrogen gas
of hydrogen gas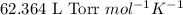
![21^oC=[21+273]K=294K](/tpl/images/0571/0032/32d52.png)