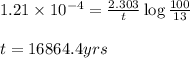Chemistry, 30.03.2020 20:15 buddyclayjohnson
Carbon-14 (14C) dating assumes that the carbon dioxide on the Earth today has the same radioactive content as it did centuries ago. If this is true, then the amount of 14C absorbed by a tree that grew several centuries ago should be the same as the amount of 14C absorbed by a similar tree today. A piece of ancient charcoal contains only 13% as much of the radioactive carbon as a piece of modern charcoal. How long ago was the tree burned to make the ancient charcoal? (The half-life of 14C is 5715 years. Round your answer to one decimal place.)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 22:30
The vapor pressure of ethanol is 1.00 × 102 mmhg at 34.90°c. what is its vapor pressure at 61.61°c? (δhvap for ethanol is 39.3 kj/mol.)
Answers: 2

Chemistry, 22.06.2019 22:30
Which of these statements best explains why space exploration should be encouraged? it prepares humans to live without oxygen. it dispel myths about objects in space. it prevents comets and asteroids from striking earth. it creates technology to absorb harmful radiations in space.
Answers: 1
You know the right answer?
Carbon-14 (14C) dating assumes that the carbon dioxide on the Earth today has the same radioactive c...
Questions










Physics, 28.07.2019 02:00

Physics, 28.07.2019 02:00




Physics, 28.07.2019 02:00





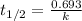
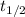 = half life of the reaction = 5715 years
= half life of the reaction = 5715 years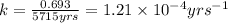
![k=\frac{2.303}{t}\log\frac{[A_o]}{[A]}](/tpl/images/0570/9587/f1041.png)
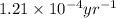
![[A_o]](/tpl/images/0570/9587/dc622.png) = initial amount of the sample = 100 grams
= initial amount of the sample = 100 grams