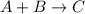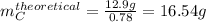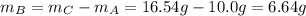Chemistry, 30.03.2020 20:11 sydneyfarrimonp9k3j4
Compound A reacts with Compound B to form only one product, Compound C, and it's known the usual percent yield of C in this reaction is 78%. Suppose 10.0 g of A are reacted with excess Compound B, and 12.9g of Compound C are successfully isolated at the end of the reaction.
1. What was the theoretical yield of C?
2. How much B was consumed by the reaction?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:00
Is this a scientific model? use complete sentences to explain why or why not. a graphic organizer showing the water cycle i need : ( asap i go it never mind
Answers: 2

Chemistry, 21.06.2019 20:30
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1
You know the right answer?
Compound A reacts with Compound B to form only one product, Compound C, and it's known the usual per...
Questions

Mathematics, 11.10.2019 18:30


History, 11.10.2019 18:30

English, 11.10.2019 18:30


Mathematics, 11.10.2019 18:30



Mathematics, 11.10.2019 18:30

Spanish, 11.10.2019 18:30

Mathematics, 11.10.2019 18:30

Mathematics, 11.10.2019 18:30


Chemistry, 11.10.2019 18:30


Mathematics, 11.10.2019 18:30


Mathematics, 11.10.2019 18:30

Mathematics, 11.10.2019 18:30