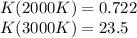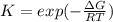The equation represents the decomposition of a generic diatomic element in its standard state. 1 2 X 2 ( g ) ⟶ X ( g ) Assume that the standard molar Gibbs energy of formation of X(g) is 5.41 kJ⋅mol − 1 at 2000. K and − 52.48 kJ⋅mol − 1 at 3000. K. Determine the value of K (the thermodynamic equilibrium constant) at each temperature. K at 2000. K = K at 3000. K =

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 23.06.2019 01:00
Which statement is true regarding the diagram of circle p? the sum of y and z must be 2x. the sum of y and z must be x. the difference of z and y must be 2x. the difference of z and y must be x
Answers: 1

Chemistry, 23.06.2019 01:30
Which of the following statements is true about energy quantization at the atomic level? electrons in the outermost orbits are the most stable. electrons in all the orbits around the nucleus have the same amount of energy. electrons in the orbit closest to the nucleus have the least amount of energy. electrons absorb or release the same amount of energy independent of the energy levels.
Answers: 1

Chemistry, 23.06.2019 04:31
Pls i will do pls imma diewhat forms white light? (4 points)a. combination of all wavelengths of ultraviolet light b. combination of all wavelengths of visible lightc. absorption of electromagnetic waves d. absorption of infrared rays
Answers: 2
You know the right answer?
The equation represents the decomposition of a generic diatomic element in its standard state. 1 2 X...
Questions

History, 04.08.2019 17:30

Mathematics, 04.08.2019 17:30




Biology, 04.08.2019 17:30

Mathematics, 04.08.2019 17:30

Mathematics, 04.08.2019 17:30

Biology, 04.08.2019 17:30

Mathematics, 04.08.2019 17:30

Mathematics, 04.08.2019 17:30

Mathematics, 04.08.2019 17:30

Mathematics, 04.08.2019 17:30

Mathematics, 04.08.2019 17:30


Mathematics, 04.08.2019 17:30

History, 04.08.2019 17:30

Mathematics, 04.08.2019 17:30


Chemistry, 04.08.2019 17:30