
Chemistry, 30.03.2020 17:01 zachtsmith2001
Be sure to answer all parts. Hydrogen iodide decomposes according to the reaction 2 HI(g) ⇌ H2(g) + I2(g) A sealed 1.50−L container initially holds 0.00623 mol of H2, 0.00414 mol of I2, and 0.0244 mol of HI at 703 K. When equilibrium is reached, the concentration of H2(g) is 0.00467 M. What are the concentrations of HI(g) and I2(g)?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 22.06.2019 01:50
7. what temperature is need to just dissolve 50 g of nh4cl in 75 g of water? '
Answers: 1


Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1
You know the right answer?
Be sure to answer all parts. Hydrogen iodide decomposes according to the reaction 2 HI(g) ⇌ H2(g) +...
Questions

Mathematics, 31.05.2021 02:20


Mathematics, 31.05.2021 02:20


Mathematics, 31.05.2021 02:20






Social Studies, 31.05.2021 02:20


Mathematics, 31.05.2021 02:20

Business, 31.05.2021 02:20



Mathematics, 31.05.2021 02:20



Mathematics, 31.05.2021 02:20

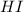 and
and 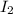 at equilibrium is, 0.0158 M and 0.00302 M respectively.
at equilibrium is, 0.0158 M and 0.00302 M respectively.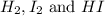
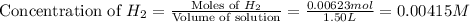
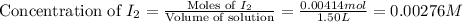
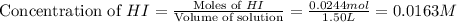
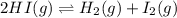
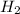 at equilibrium = 0.00467 M
at equilibrium = 0.00467 M

