Chemistry, 30.03.2020 04:55 ianball025
A sample of flammable liquid is placed into an enclosed cylinder which is then fitted with a movable piston. Initially the cylinder contains a volume of 1.60 L. The sample is ignited producing gas and releasing 401.5 J of energy. To what volume will the cylinder expand to if it must expand against a pressure of 729.8 mmHg. Assume all the energy released is converted to work used to push the piston?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
If you have 5.25 grams of methane (ch4), how many grams of co2 will you produce ?
Answers: 1

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1

Chemistry, 22.06.2019 16:00
About 3% of the water on earth is freshest. only about 40% of that freshwater is available for human use. why is so much freshwater unavailable for human use?
Answers: 2

Chemistry, 23.06.2019 00:30
The molecular weight of carbon dioxide, co2, is 44.00 amu, and the molecular weight of nitrous dioxide, no2, is 46.01 amu, so no2 diffuses co2
Answers: 2
You know the right answer?
A sample of flammable liquid is placed into an enclosed cylinder which is then fitted with a movable...
Questions


Mathematics, 15.10.2020 03:01


Social Studies, 15.10.2020 03:01

Chemistry, 15.10.2020 03:01

Mathematics, 15.10.2020 03:01

Mathematics, 15.10.2020 03:01

Geography, 15.10.2020 03:01


Mathematics, 15.10.2020 03:01




Mathematics, 15.10.2020 03:01

History, 15.10.2020 03:01

Mathematics, 15.10.2020 03:01



Mathematics, 15.10.2020 03:01

Mathematics, 15.10.2020 03:01

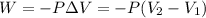
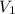 = initial volume = 1.60 L
= initial volume = 1.60 L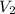 = final volume = ?
= final volume = ?